


1 / 5













| Technical Category | Detailed Specifications |
|---|---|
| Machine Type | Ethylene Oxide sterilizers / Sterilization chambers |
| Chamber Capacity | 1 to 100 m3 |
| Chamber Construction | Rectangular, 1 to 13 pallets (80x120cm) |
| Chamber Material | Stainless steel grade 304 or 316 |
| Heating System | Double jacket with hot water circulation (Integrated tank) |
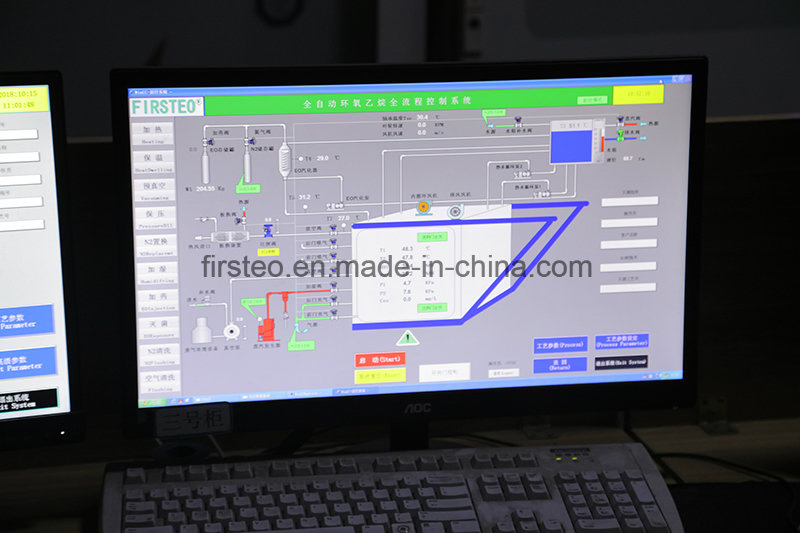
| PLC & Controls | SIEMENS Programmable Logic Controller |
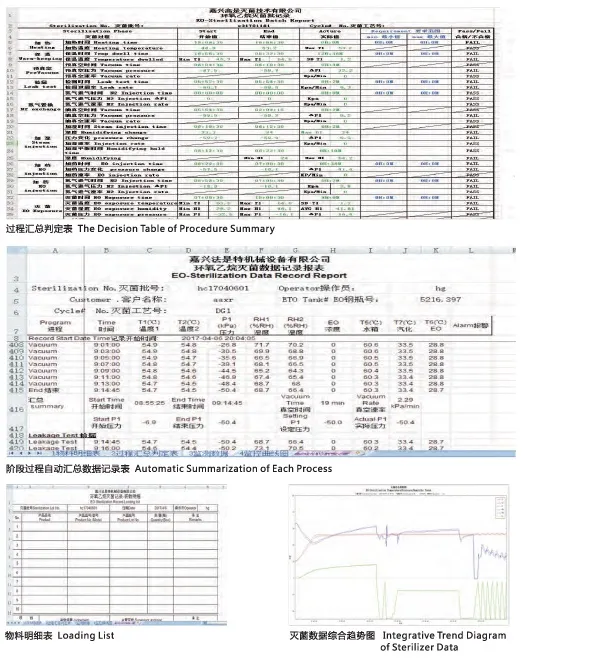
| Data Processing | Automatic/Manual control, Graphic recorder (Temp, Humidity, Pressure) |
| Power Supply | 380V AC, 50Hz or 60HZ |